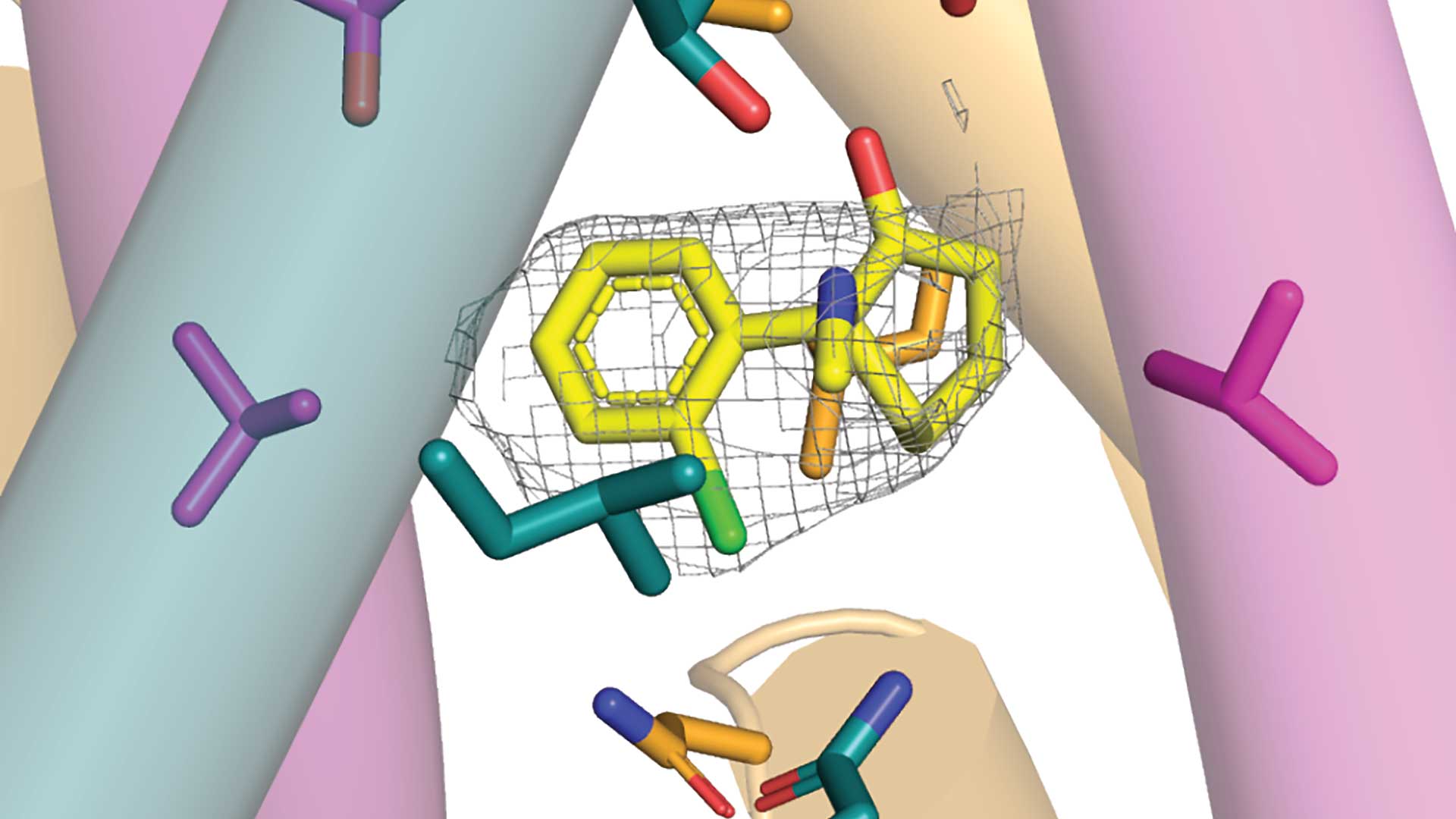
It’s been theorized that ketamine works by blocking a brain receptor called GluN1-2B-2D. Here, we see one of the many ways the drug’s chemical molecules (yellow) can bind to a specific part (the gray web) of the brain receptor. Ketamine has received a Hollywood makeover. It used to be known as a rave drug (street name special K) and cat anesthetic. However, in recent years, some doctors have prescribed ketamine to treat conditions from post-traumatic stress disorder to depression. “The practice is not without controversy,” notes Cold Spring Harbor Laboratory (CSHL) Professor Hiro Furukawa .
‘Should we give a hallucinogen to patients in compromised mental states?’ wonder ketamine’s skeptics. The controversy came to a head in 2024 following the death of Matthew Perry. The popular TV actor, best known as Chandler on NBC’s Friends , died from a ketamine overdose. One person charged in connection with Perry’s death was the doctor who’d prescribed him ketamine for depression and anxiety. This 3D animation illustrates the tension-and-release mechanism that controls how brain receptor GluN1-2B-2D opens and closes its ion channel pore.
“Even putting this aside, many questions remain regarding how ketamine affects the brain,” says Furukawa. “It’s been suggested for over a decade that the drug blocks a specific kind of NMDA receptor (NMDAR), called GluN1-2B-2D.” There was one big problem with this theory. Scientists weren’t quite sure that GluN1-2B-2D existed. A new study from the Furukawa lab shines much-needed light on the situation.
In a paper published in the journal Neuron , Furukawa and postdoc Hyunook Kang prove that GluN1-2B-2D does exist in the mammal brain. They then reconstruct a human version of GluN1-2B-2D. They don’t stop there. Using electron cryo-microscopy (cryo-EM), they capture GluN1-2B-2D in action. The neuroscientists identify the tension-and-release mechanism that controls GluN1-2B-2D movements. They can now see how this mysterious NMDAR opens and closes its ion channel pore. And they go another step further. They reveal several ways ketamine may bind to GluN1-2B-2D. Furukawa and colleagues zoomed in on ketamine’s binding site within GluN1-2B-2D, revealing four ways the drug could close off the channel. A series of stunningly detailed […]

Get Creative with Cannabis: The Ultimate Guide to Painting and Pottery Workshops
Creative with Cannabis Cannabis and creativity go hand in hand—especially